The Microfluidics Group is developing high-throughput and high-content assay platforms for many aspects of bioenergy and biomanufacturing research at JBEI. This involves using novel microfluidic devices, lab automation, and AI/ML models to advance various biotechnology processes such as electroporation-based microbial transformation. The goal of the group is to develop cutting-edge microscale technologies for robust biofuel and bioproduct development and to generate high-quality datasets for training next-gen AI/ML models to make synthetic biology more predictable.
Projects
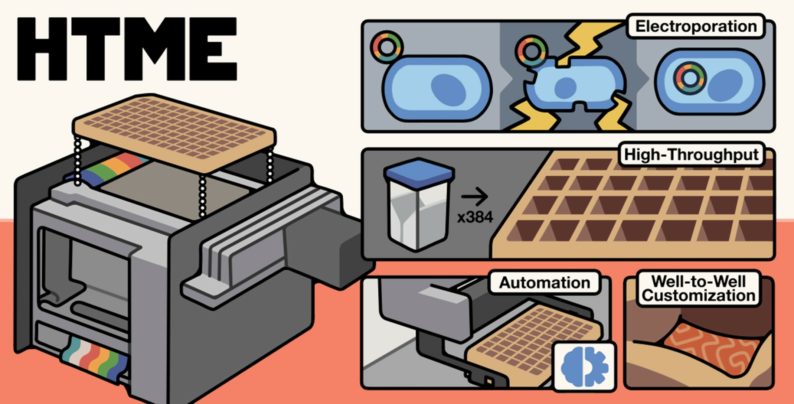
- A fully automated High-Throughput Microfluidic Electroporation (HTME) Platform performing 384 sub-microliter volume electroporation experiments in parallel.
- Using CRISPR-MAGE motifs to edit biofuel protein pathways in multiple species of microorganisms.
- Self-Driving Laboratory (SDL) automation process development.
- Assay development at the interface of droplet microfluidics and mass spectrometry to streamline the discovery and optimization of biomanufacturing pathways.
- Development of a method for Next-Generation DNA Synthesis.
- A microfluidic cell free platform utilizing an array of digital microfluidic (DMF) electrodes to quantify the activity of multi-component protein pathways.
Featured Media
Microfluidic Device Mixes And Matches DNA For Synthetic Biology
Publications
- “High-Throughput Microfluidic Electroporation (HTME): A Scalable, 384-Well Platform for Multiplexed Cell Engineering”, Bioengineering, (2025)
- “Perspectives for self-driving labs in synthetic biology”, Current Opinion in Biotechnology (2023)
- “Scalable and automated CRISPR-based strain engineering using droplet microfluidics”, Microsyst Nanoeng, (2022)
- “A machine learning Automated Recommendation Tool for synthetic biology”, Nat Communications, (2020)
- “Predictive engineering and optimization of tryptophan metabolism in yeast through a combination of mechanistic and machine learning models”, Nature Communications, (2020)
- “A droplet-to-digital (D2D) microfluidic device for single cell assays”, Lab Chip (2015)
- “A Versatile Microfluidic Device for Automating Synthetic Biology”, ACS Synth Biol (2015)
- “Pressure stabilizer for reproducible picoinjection in droplet microfluidic systems”, Lab Chip (2014)
- “Versatile on-demand droplet generation for controlled encapsulation”, Biomicrofluidics (2014)
- “Rapid Kinetic Characterization of Glycosyl Hydrolases Based on Oxime Derivatization and Nanostructure-Initiator Mass Spectrometry (NIMS)”, ACS Chem Biol (2014)
- “Droplet-based microfluidic platform for heterogeneous enzymatic assays”, Lab Chip (2013)
- “A universal flow cytometry assay for screening carbohydrate-active enzymes using glycan microspheres”, Chem Communications (2013)
- “Acoustic deposition with NIMS as a high-throughput enzyme activity assay”, Analytical and Bioanalytical Chemistry (2012)
- “Nanostructure-initiator mass spectrometry (NIMS) for the analysis of enzyme activities”, Curr protoc chem biol (2012)
- “Encoding substrates with mass tags to resolve stereospecific reactions using Nimzyme”, Rapid Communications in Mass Spectrometry (2012)
- “Colloid-based multiplexed screening for plant biomass-degrading glycoside hydrolase activities in microbial communities”, Energy and Environmental Science (2011)
- “Microfluidic Glycosyl Hydrolase Screening for Biomass-to-Biofuel Conversion”, Analytical Chemistry (2010)
Intellectual Property
- Microfluidic Platform for Synthetic Biology Applications
- Contactless liquid loading to microfluidic devices
- Apparatus and methods for increased transformation efficiency for electroporation of microorganisms
- Method and device for tracking and manipulation of droplets
- Microfluidic platform for synthetic biology applications