The Science
Breaking down the components of lignocellulosic biomass into fermentable sugars that can be converted into biofuels and bioproducts — a process known as saccharification — remains a challenge due to the fact that biomass is naturally resistant to biological degradation and industrial processing. Enzymatic saccharification, in which enzymes break down cellulose in lignocellulosic biomass, has been used to overcome this resistance. Many microorganisms, such as certain fungi, secrete mixtures of enzymes that can be used for saccharification. These enzyme mixtures contain lytic polysaccharide monooxygenases (LPMOs), which contribute to saccharification by cutting through cellulose chains. Though LPMOs were discovered over 10 years ago, there are still several unanswered questions about their mechanisms and how they can be optimized to process lignocellulosic biomass.
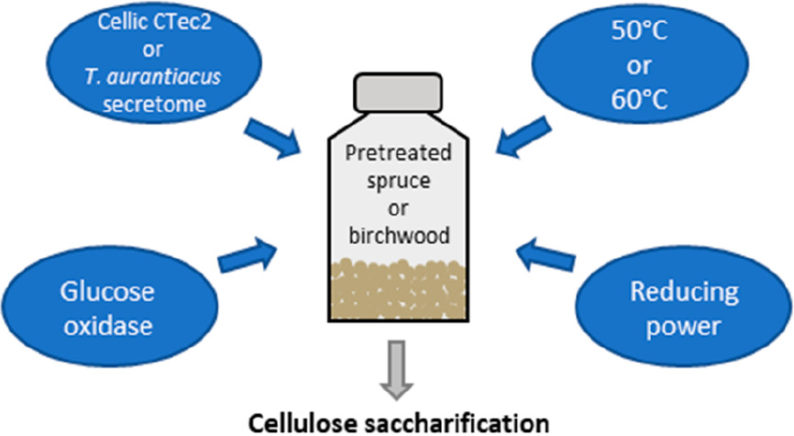
The temperature limitations of enzymes present another challenge in biomass processing. Research has shown that performing saccharification at high temperatures could be beneficial. This has made the fungus T. aurantiacus of particular interest, as it can secrete an LPMO-rich enzymatic mixture that functions at high temperature.
The Impact
Researchers at the Joint BioEnergy Institute, in collaboration with the Norwegian University of Life Sciences, set out to gain a better understanding of how to optimize enzymatic saccharification of biomass, focusing on how LPMOs play a role. They were able to compare the performance of a commercial enzyme with the LMPO-rich secretome of T. aurantiacus. They found there are many factors that impact the effectiveness of enzymatic saccharification involving LMPOs. Understanding these factors could help optimize saccharification processes.
Summary
The researchers compared the T. aurantiacus secretome with the performance of Cellic CTec2, a commercial enzyme used for biomass degradation. Specifically, they compared how sensitive each enzymatic mixture was to the presence of lignin in pretreated biomass. On a lignin-poor substrate, Cellic CTec2 performed better at a lower temperature, but the T. aurantiacus secretome was able to match the Cellic CTec2 performance at higher temperature. On lignin-rich substrate, Cellic Ctec2 performed better than the T. aurantiacus secretome. In all cases, the T. aurantiacus LPMO was highly active and was shown to be a key component of the enzymatic mixture. These findings suggest that the native LPMO-rich T. aurantiacus secretome has activity that is comparable to commercial enzymatic cocktails and could be useful for saccharification in lignin-poor conditions.
Publication Citation:
Heidi Østby, Anikó Várnai, Raphael Gabriel, Piotr Chylenski, Svein J. Horn, Steven W. Singer, and Vincent G. H. Eijsink (2022) “Substrate-Dependent Cellulose Saccharification Efficiency and LPMO Activity of Cellic CTec2 and a Cellulolytic Secretome from Thermoascus aurantiacus and the Impact of H2O2-Producing Glucose Oxidase.” ACS Sustainable Chemistry & Engineering. DOI: 10.1021/acssuschemeng.2c03341