The Science
Enzymes are substances that cause chemical reactions. Certain types of enzymes, such as polyketide synthases and nonribosomal peptide synthetases, have the ability to shuffle their parts, allowing them to produce new chemicals. If scientists can understand how these enzymes shuffle their parts, they can understand how to use them to synthesize millions of molecules, such as pharmaceuticals and biofuels. However, engineering these enzymes is difficult because scientists don’t fully understand how they work. In this study, researchers describe the improvements they made to an online tool, ClusterCAD, that helps scientists design and test engineered variants of enzymes.
The Impact
Engineering these key enzymes to produce chemicals has become a major focus in synthetic biology. ClusterCAD is the only available tool that helps researchers design these enzymes for synthetic biology applications. Researchers made new improvements to the tool, including an expanded database, powerful search tools, and helpful new features within the interface. These updates will expand the types of chemicals that scientists can access with enzymes engineered by ClusterCAD.
Summary
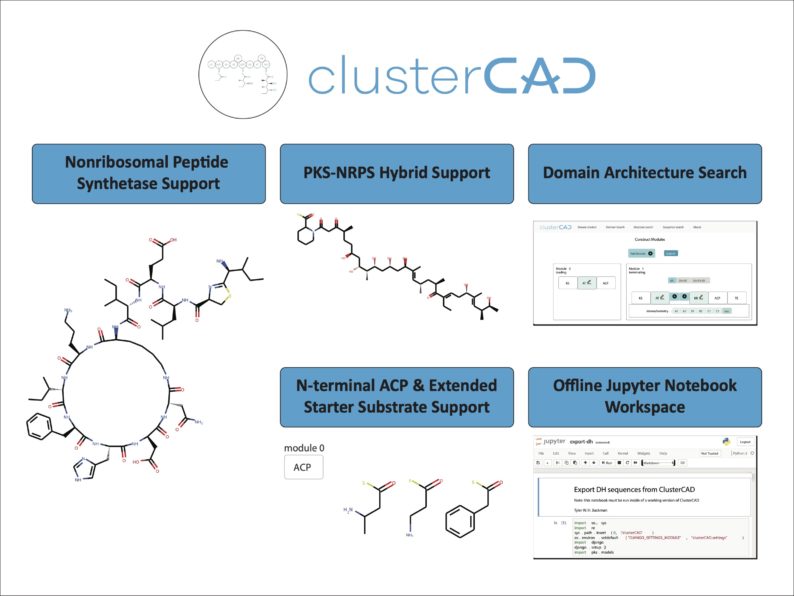
Researchers made several improvements to the existing ClusterCAD tool. ClusterCAD is a free online platform that simplifies the process of designing and testing engineered enzyme variants for synthetic biology applications. The tool allows users to browse polyketide synthase (PKS) and nonribosomal peptide synthetase (NRPS) gene clusters, which are groups of genes that encode pathways that can produce chemicals.
With this update, users can now design NRPS clusters within the platform. The researchers also expanded the platform’s database of clusters to be more comprehensive and informative. Other updates provide users with more high-quality clusters available for study, expanded starting points for PKS and NRPS engineering, and improved database versatility and search tools. These updates allow users to explore more chemicals that can be produced through PKS and NRPS engineering.
Funding
Funding was provided by the Department of Energy’s (DOE) Office of Energy Efficiency and Renewable Energy (EERE) and Bioenergy Technologies Office. Funding was also provided by the DOE Joint BioEnergy Institute, which is supported by the DOE Office of Science, Office of Biological and Environmental Research. Funding was also provided by the Philomathia Foundation.
Publications
Tao, X.B. et al., ClusterCAD 2.0: an updated computational platform for chimeric type I polyketide synthase and nonribosomal peptide synthetase design. Nucleic Acids Research 1(2022). [DOI: 10.1093/nar/gkac1075]
This science highlight was originally published on Energy.gov