– By Irina Silva
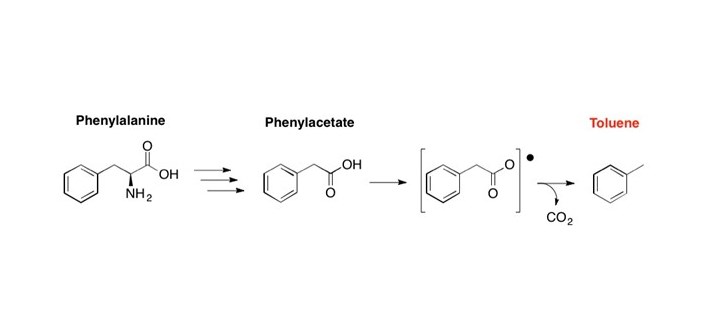
In a Scientific Reports (Nature) paper entitled “In vitro characterization of phenylacetate decarboxylase, a novel enzyme catalyzing toluene biosynthesis in an anaerobic microbial community”, researchers at JBEI investigated an enzyme that could enable first-time biochemical production of the widely used octane booster, toluene.
Twenty years ago, it was reported that bacteria in anoxic environments could produce the important industrial chemical, toluene, a widely used octane booster with a global market of 29 million tons per year. Since that time, the enzyme responsible for catalyzing the intriguing and potentially useful reaction of toluene biosynthesis has remained a mystery. If biotechnology could harness this enzyme, it could promote sustainability by offsetting the enormous volume of petroleum-derived toluene with biochemically produced toluene made from a renewable resource, such as lignocellulosic biomass.
Researchers at JBEI, led by Harry Beller (Director of Biofuel Pathways at JBEI), and colleagues at DOE’s Joint Genome Institute (JGI), have made significant progress in elucidating the nature of the toluene synthase enzyme. After establishing an anaerobic toluene-producing microbial community that was inoculated with sewage sludge, they investigated the proteins being expressed in this community using a combination of protein separation technologies, custom mass spectrometric techniques, and metagenomic and metaproteomic analysis. Although the exact identity (i.e., gene/protein sequence) of the novel enzyme was not yet obtained, the detailed characteristics of the enzymatic reaction that were reported will facilitate its identification. The researchers are confident that they will soon be completing the discovery of the toluene synthase enzyme, i.e., determining its sequence, thus enabling first-time production of an aromatic hydrocarbon for biofuels.